Powered by the HAPPE® platform, INTEGRATE®-C is the first interbody fusion cage to offer cancellous porosity with fully integrated hydroxyapatite through the entire implant height, combined with radiolucency and bone-like mechanical properties.

Integrate (verb):
to combine two or more things in order to become more effective

Porous and Dense Material
INTEGRATE® has porous and dense material designed for optimum biomechanics in load bearing and healing.
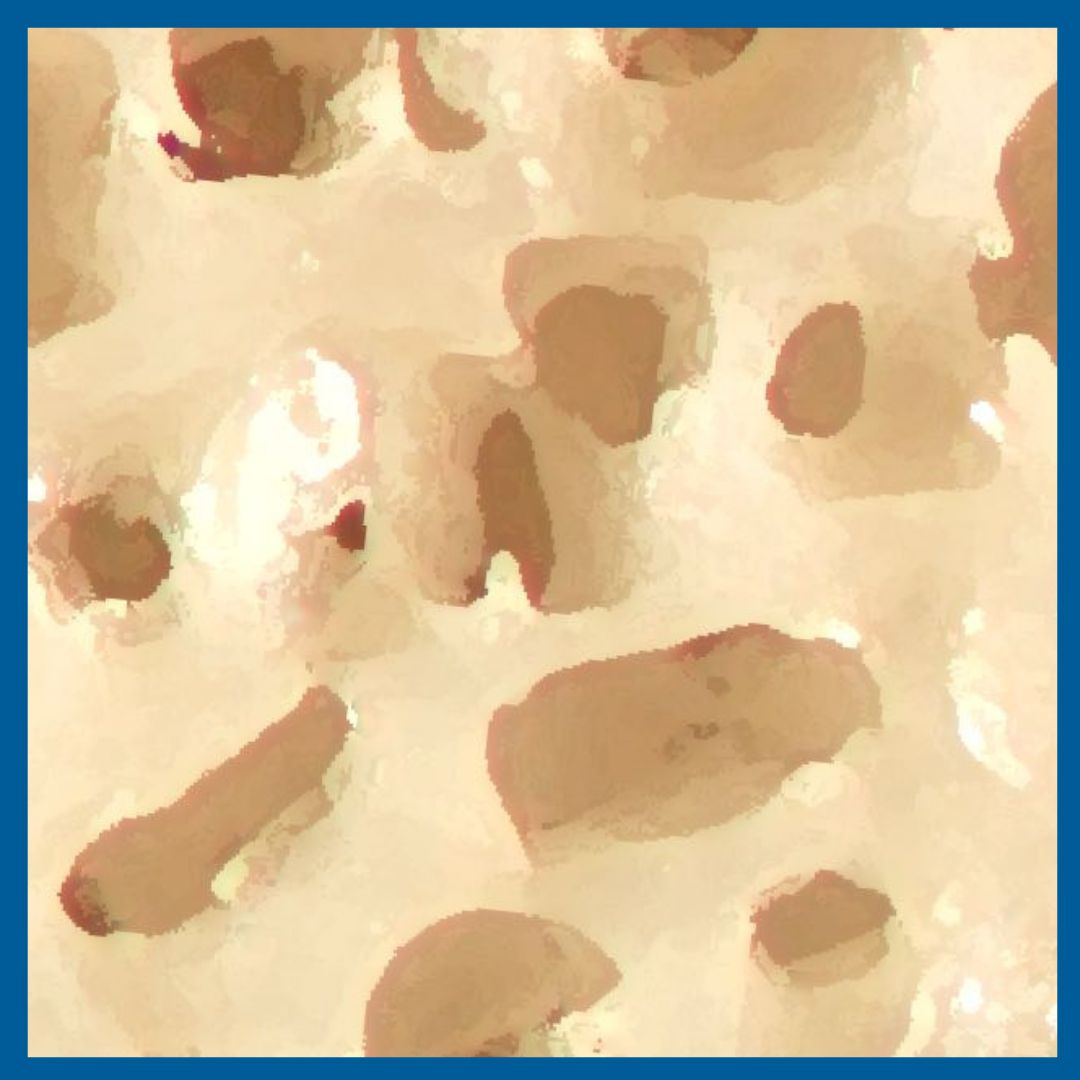
Interconnected Cancellous Porosity
INTEGRATE® has interconnected cancellous porosity for bone ingrowth endplate-to-endplate.
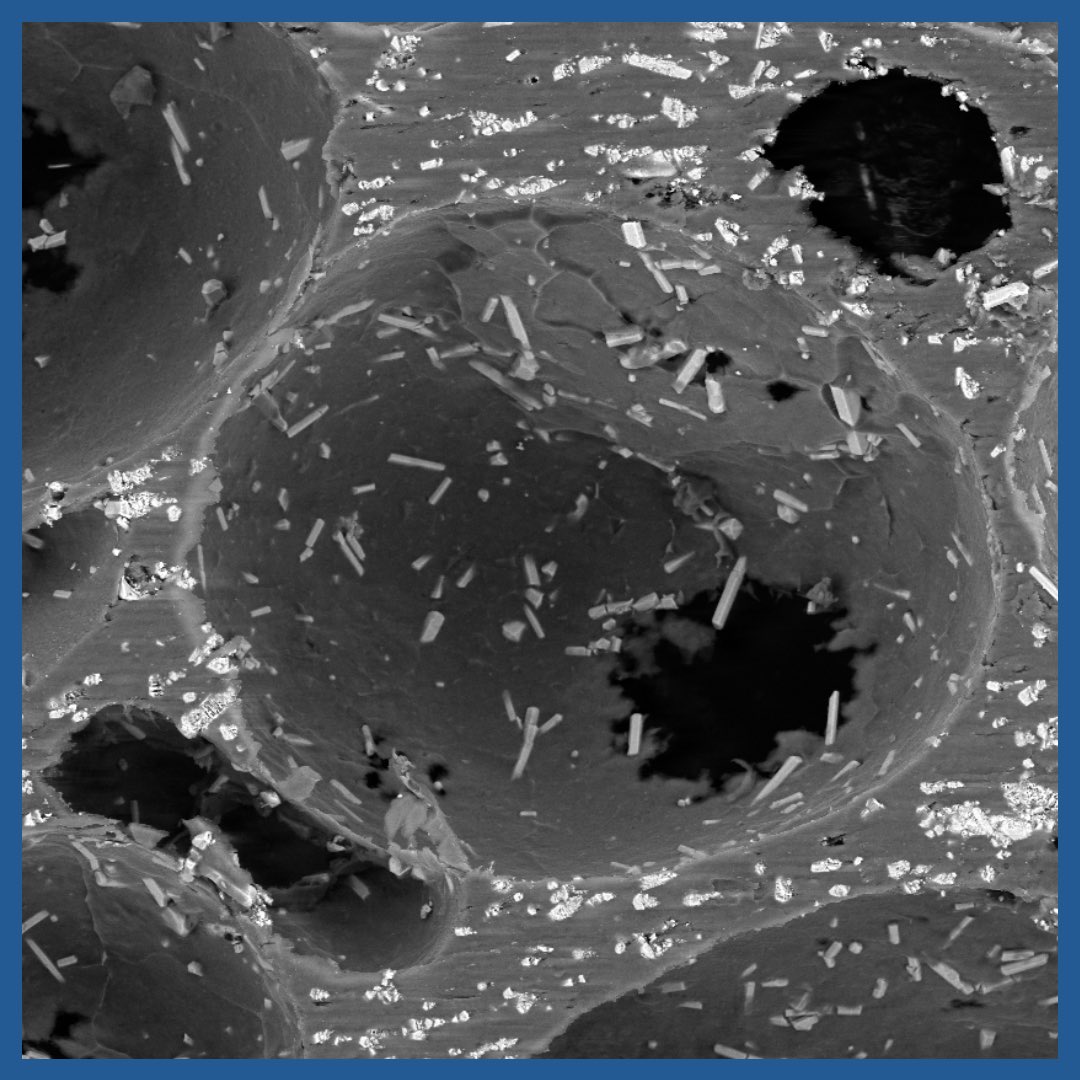
Hydroxyapatite Exposed on Surfaces
INTEGRATE® has hydrophilic surfaces impregnated with hydroxyapatite, for bone ongrowth.
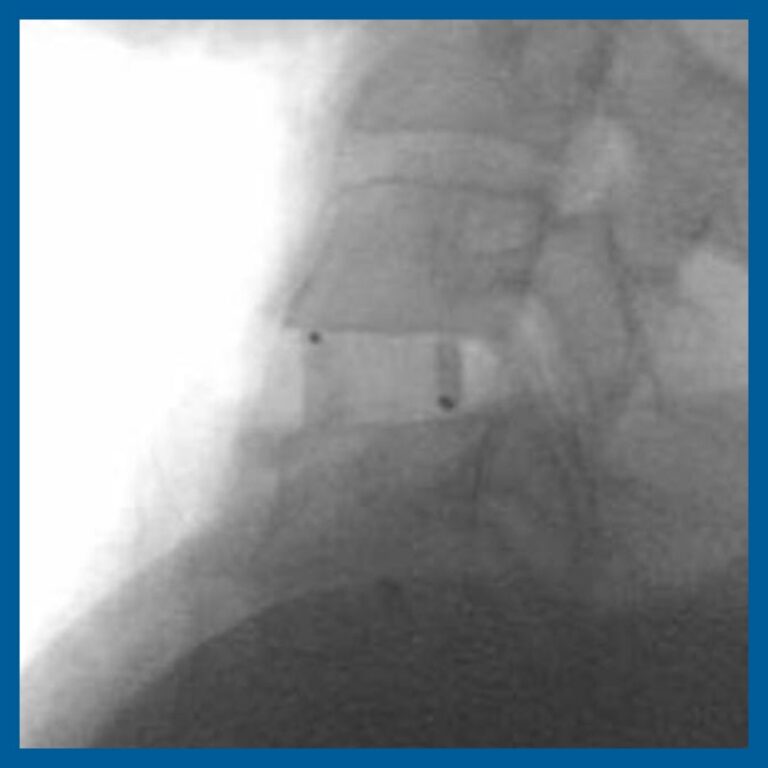
Radiolucent and Radiovisible
INTEGRATE® is both radiolucent and radiovisible for superior post-operative imaging.
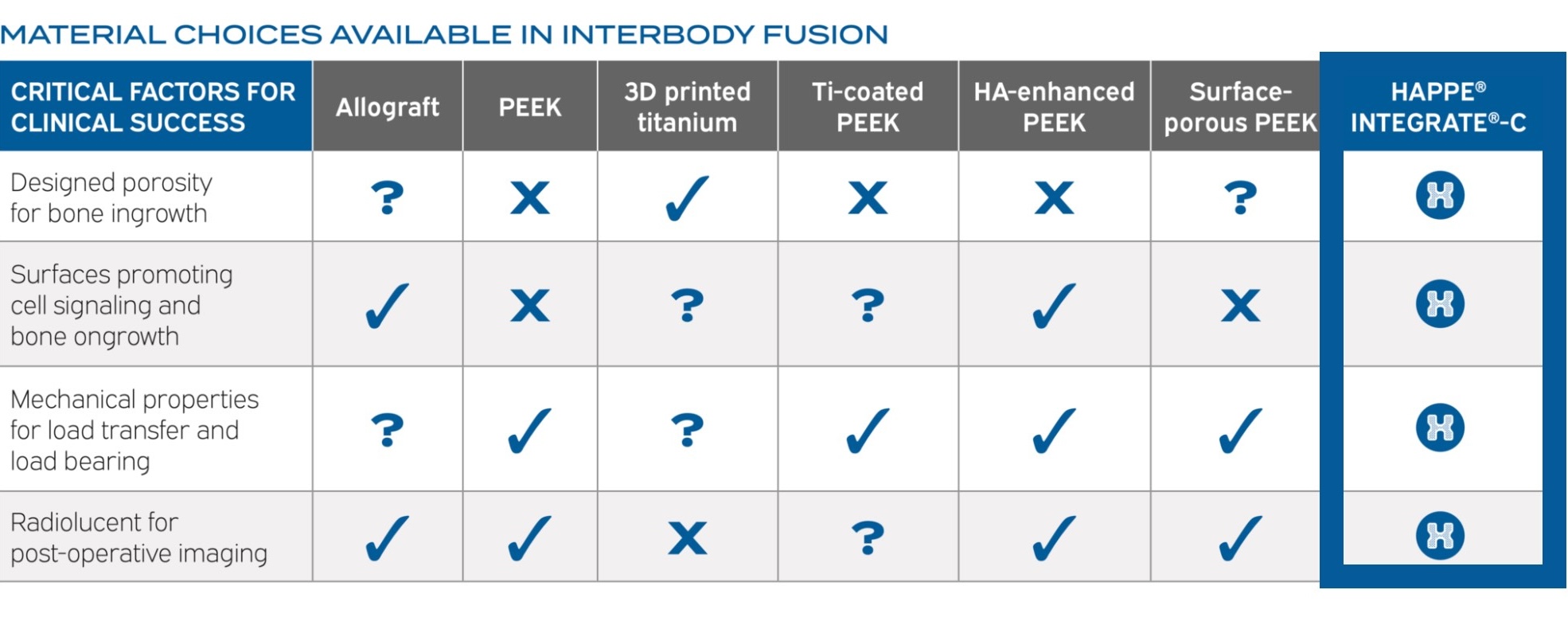
MATERIAL CHOICES AVAILABLE IN INTERBODY FUSION
Bone ingrowth into HAPPE implants was confirmed in an ovine cortical bone defect model.*
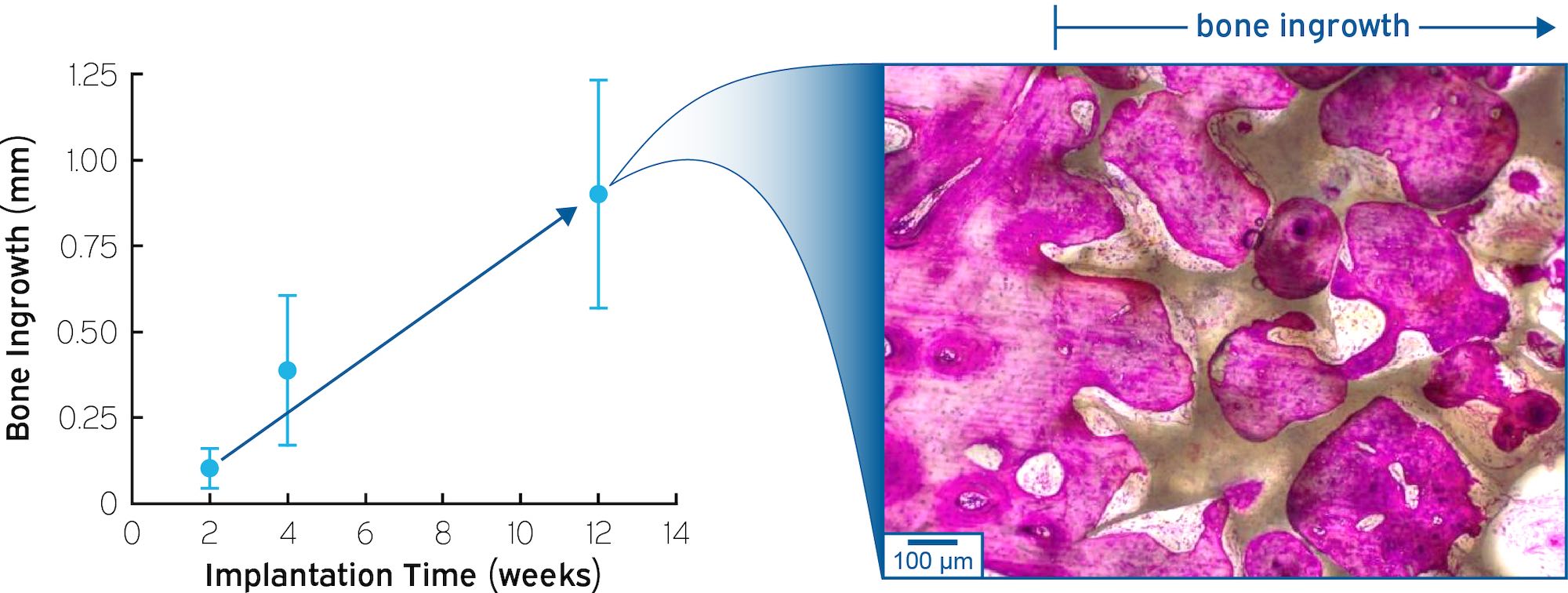
*R.K. Roeder, J.F. Nagle and D. Snell, “Translating Porous and Bioactive PEEK to Interbody Spinal Fusion Implants,” Society for Biomaterials Annual Meeting, 2021. University of New South Wales – Surgical & Orthopaedic Research Laboratories – Evaluation of osteointegration in a large animal model. Note: the porous structure of the HAPPE® material in the INTEGRATE®-C Fusion Device has been modified from that used for this ovine study with the intent to further increase pore interconnectivity for bone in-growth. In vitro performance or animal studies may not be representative of clinical performance.
The INTEGRATE-C Fusion System
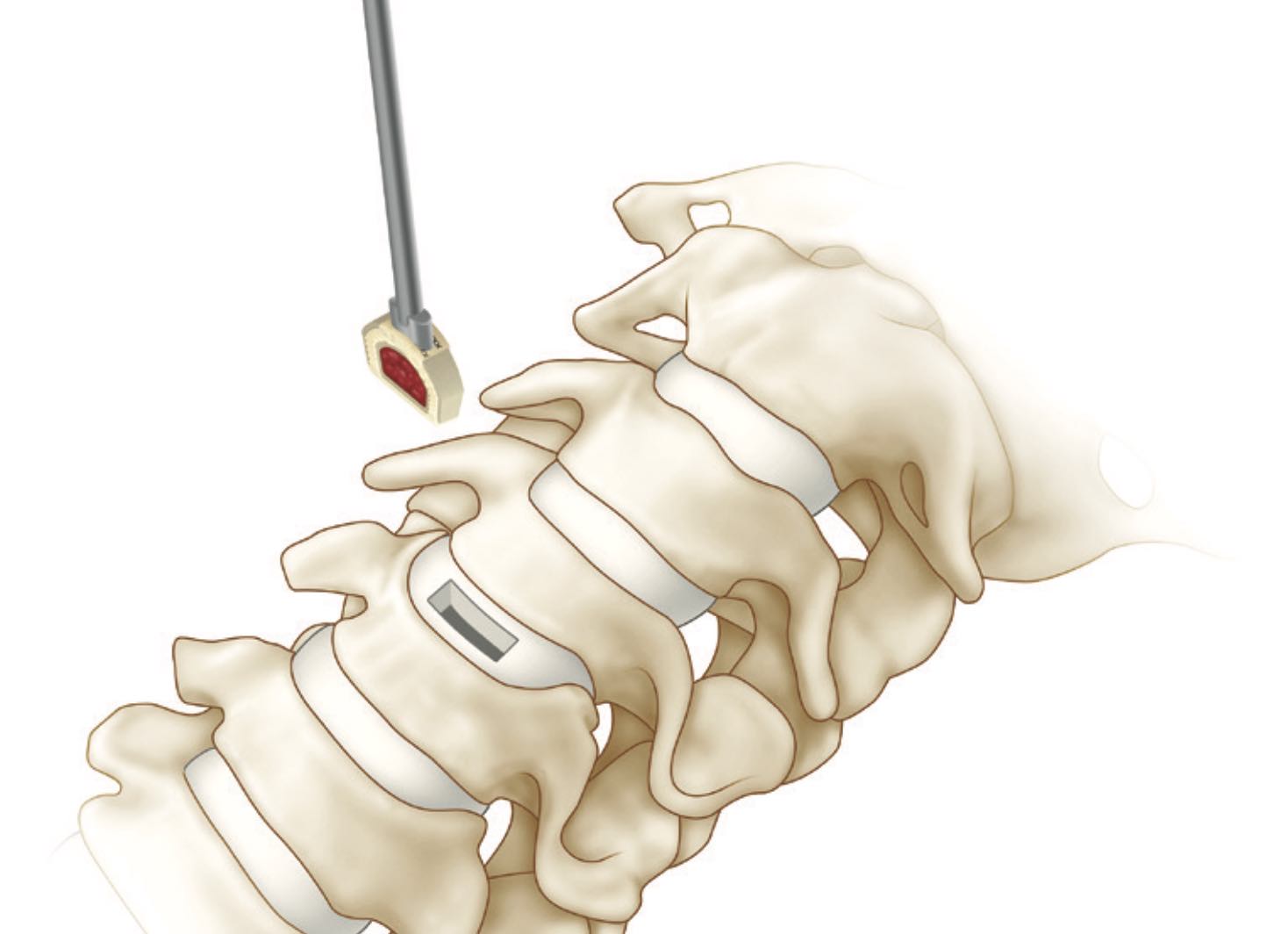
The INTEGRATE®-C Fusion Device is made of a single, continuous piece of hydroxyapatite impregnated polyetheretherketone polymer.
The INTEGRATE®-C Fusion Device is monolithic with porous regions derived directly from the implant body, not a sintered or otherwise additive coating, and extended through the device.
The device is available in a variety of footprints, lordosis and heights to accommodate variations in the individual pathology and anatomy of the patient. The superior and inferior surfaces of the device contain a pattern of teeth to provide for initial stability. Radiopaque markers are placed in the device to aid in determining the location of the implant intra- and post-operatively.
Features and Benefits
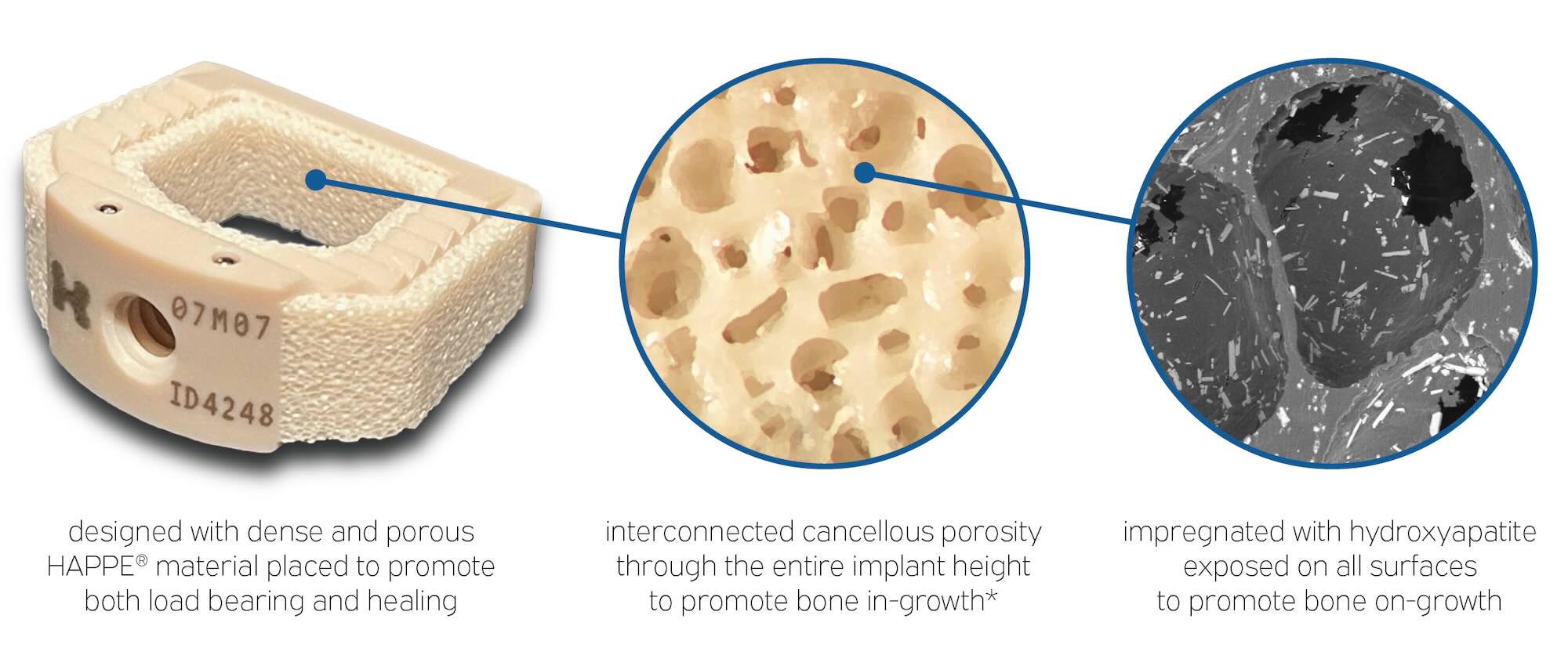
- Made with patented HydroxyApatite Porous PolyEtheretherketone (HAPPE®)
- Large graft space lined with porous HAPPE material for endplate-to-endplate osteointegration
- Dense structural element for load bearing and impaction
- Exposed hydroxyapatite crystals throughout the implant for hydrophilic surfaces
- Implant sizes include 15 x 12, 17 x 14 and 20 x 15 mm with 7 or 10 degree lordosis for anatomic fit
- Tapered design for easy insertion and aggressive teeth for preventing expulsion
- Tantalum markers for radiographic visualization
- Radiolucent for post-operative assessment of fusion
Stay Connected with HAPPE
Sign up to receive the latest news and updates from HAPPE.

